
These include an increased risk of mutation, retention of somatic epigenetic memory, potential for immunogenicity, and altered functional characteristics of differentiated phenotypes 12, 13, 14, 15. However, conventional methods for the induction and maintenance of pluripotent state introduce a myriad of additional biological obstacles to effective application in stem-cell-derived therapeutics. Utilization of hiPSCs as an alternative cell source effectively overcomes the legal, ethical, and moral barriers associated with the use of embryonic or epiblast stem cells 11. However, harvesting the cells requires destruction of the developing embryo. Naturally occurring populations of these cells are found within an early-stage embryonic blastocyst and can be isolated and expanded in vitro to generate a population of self-renewing human embryonic stem cells (ESCs) 10. Human pluripotent stem cells are promising candidates for therapeutic applications given their proliferative potential and capacity to differentiate into any cell type within the primary germ layers of an adult organism 9. This paper aims to explore the challenges and potential strategies to overcome obstacles faced in controlling pluripotent and differentiated phenotype in the context of functionality, efficacy, and safety throughout hiPSC biomanufacturing. These advancements will require the integration of process systems, novel analytical technologies, and computational methods throughout development and optimization. Utilization of human induced pluripotent stem cells (hiPSCs) and their derivatives for drug discovery, cell therapy, and disease modeling present a potentially invaluable resource, but existing limitations to their use include conceptual biases, technological limitations, process heterogeneity, rudimentary control over cell fate, and substantial cost 7, 8.Īddressing these limitations and obstacles in the development of stem-cell-based therapeutics includes the incorporation of technologies and methods capable of continuous monitoring and assessment of phenotype throughout the bioprocess, in conjunction with process control, standardization, and automation of protocols as they are developed. Clinical outcomes are dependent on biological function of the product, with quality hindered by obstacles such as a lack of reproducibility and robustness for scale-up and scale-out 6. 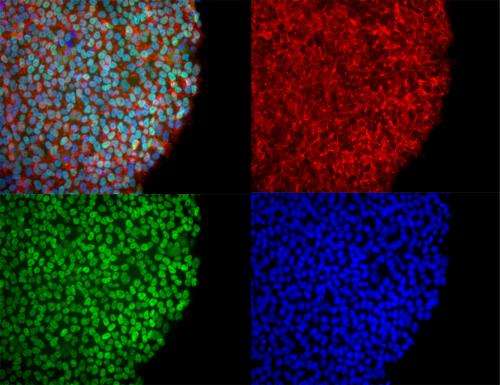
Coupled with in-process heterogeneity and the evolving regulatory framework surrounding assessment of stem-cell-derived therapeutics, it is difficult to develop scalable, robust processes that are strictly standardized and economically viable 4, 5. However, optimization of manufacturing protocols is resource-intensive and costly, with characterization of phenotype being laborious and discontinuous. Modern bioprocessing technology and protocols have developed to the point of producing functional, clinically relevant numbers of pluripotent stem cells for use as cell and tissue source material 1, 2, 3. This overarching approach is thus crucially important in realizing the potential of hiPSC biomanufacturing for transformation of regenerative medicine and the healthcare industry. Coupling complete datasets with relevant computational methods will contribute significantly to model development and automation in achieving process robustness. This includes integrating process data with biological characteristic and functional assessment data to model the interplay between variables in the search for global optimization strategies. It is argued herein that one of the major challenges currently faced in improving the robustness of routine stem-cell biomanufacturing is in utilizing continuous, meaningful assessments of molecular and cellular characteristics from process to application. The complexity and volatility of hiPSCs present several bioprocessing challenges, where the goal is to generate a sufficiently large, high-quality, homogeneous population for downstream differentiation-the derivatives of which must retain functional efficacy and meet regulatory safety criteria in application. However, while many promising therapeutic approaches are being tested in pre-clinical studies, hiPSC-derived products currently account for a small fraction of active clinical trials. The translation of laboratory-scale bioprocess protocols and technologies to industrial scales and the application of human induced pluripotent stem cell (hiPSC) derivatives in clinical trials globally presents optimism for the future of stem-cell products to impact healthcare.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
